Delegated Regulation (EU) 2026/314: expansion of the list of scheduled substances for drug precursors
The European Union has adopted Commission Delegated Regulation (EU) 2026/314 of 9 February 2026, published on 28 April 2026.
This regulation amends Regulation (EC) No 273/2004 and Regulation (EC) No 111/2005 with regard to the inclusion of new drug precursors in the list of scheduled substances.
Specifically, 9 new chemical substances are added to Category 1, the strictest level of control within the European regulatory framework.
What does Category 1 for drug precursors imply?
Inclusion in Category 1 entails highly stringent control. These substances require:
Activity licenses
Detailed operator registration
Specific import and export permits
Comprehensive monitoring of the entire supply chain
In addition, their classification facilitates the work of national authorities in combating the illegal manufacture of drugs.
From the entry into force of the regulation, these substances will be considered “precursors” according to the definition established in Framework Decision 2004/757/JHA, allowing minimum criteria to be set for defining offences related to their trafficking.
List of newly scheduled substances
The substances added are:
i) 3′-chloropropiophenone
ii) 2-bromo-3′-chloropropiophenone
iii) 3′-methylpropiophenone
iv) 2-bromo-3′-methylpropiophenone
v) 4′-methylpropiophenone
vi) 2-bromo-4′-methylpropiophenone
vii) 4′-chloropropiophenone
viii) 2-bromo-4′-chloropropiophenone
ix) phenyl-2-nitropropene
Relation to synthetic drugs
These substances act as precursors to various synthetic drugs:
3′-chloropropiophenone and 2-bromo-3′-chloropropiophenone → 3-CMC (clofedrone)
3′-methylpropiophenone and 2-bromo-3′-methylpropiophenone → 3-MMC
4′-methylpropiophenone and 2-bromo-4′-methylpropiophenone → 4-MMC (mephedrone)
4′-chloropropiophenone and 2-bromo-4′-chloropropiophenone → 4-CMC
Phenyl-2-nitropropene → amphetamine
The substances 3-CMC, 3-MMC, 4-CMC and 4-MMC are stimulants belonging to the group of synthetic cathinones and are regulated at both European and international level due to their risks to public health.
Risk assessment and legal uses
The European Commission tasked the European Union Drugs Agency (EUDA) with assessing these substances.
According to the EUDA:
They can be used in the illegal manufacture of drugs through simple processes
They require basic equipment and limited technical expertise
Their seizures have increased in recent years
However, they also have legitimate uses, for example:
Production of bupropion (treatment for depression and smoking cessation)
Manufacture of tolperisone (a muscle relaxant)
Production of amphetamine-derived medicines
Use as reference standards in analytical laboratories
Strengthening regulatory control
The Regulation states that:
“competent national authorities reported an increase in seizures of the nine non-scheduled substances”
It also indicates that, from its entry into force, these substances must be considered “precursors” for the purposes of European criminal law.
Article 1 states that Annex I to Regulation (EC) No 273/2004 is amended in accordance with Annex I of this Regulation.
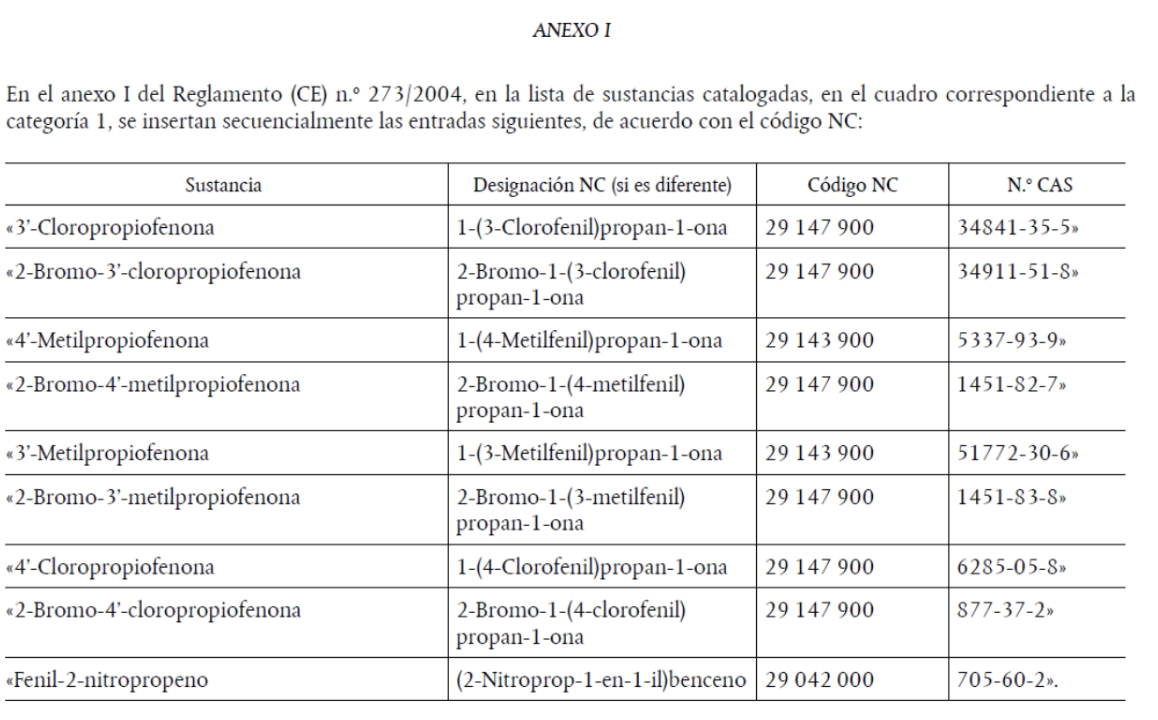
Article 2 provides that the Annex to Regulation (EC) No 111/2005 is also amended in accordance with Annex II of this Regulation.
Accordingly, this Delegated Regulation (EU) 2026/314 shall enter into force twenty days after its publication and shall apply from 18 September 2026, in order to allow the affected economic operators sufficient time to adapt their procedures and obtain the required authorisations.
Legislative amendments introduced
The Regulation establishes:
Article 1: amendment of Annex I to Regulation (EC) No 273/2004
Article 2: amendment of the Annex to Regulation (EC) No 111/2005
Entry into force and application
Delegated Regulation (EU) 2026/314:
Will enter into force 20 days after its publication
Will apply from 18 September 2026
This period allows affected economic operators to adapt their procedures and obtain the required authorisations.
If you wish to consult the full text of the regulation, you can access it here
How eQgest helps you with regulatory compliance
At eQgest, we help you efficiently manage compliance with complex regulations such as Delegated Regulation (EU) 2026/314.
Contact us to find out how we can help you comply with drug precursor regulations safely and efficiently.
Receive regulatory updates straight to your inbox
Newsletter with updated chemical regulations, regulatory news from the sector, and upcoming webinars — carefully selected for professionals like you.


